Base-Catalyzed Aryl-B(OH)2 Protodeboronation Revisited: From
$ 37.00
-
By A Mystery Man Writer
-
-
4.9(752)

Product Description

Base-Catalyzed Aryl-B(OH)2 Protodeboronation Revisited: From Concerted Proton Transfer to Liberation of a Transient Aryl Anion

PDF] A mechanistic proposal for the protodeboronation of neat boronic acids: boronic acid mediated reaction in the solid state.
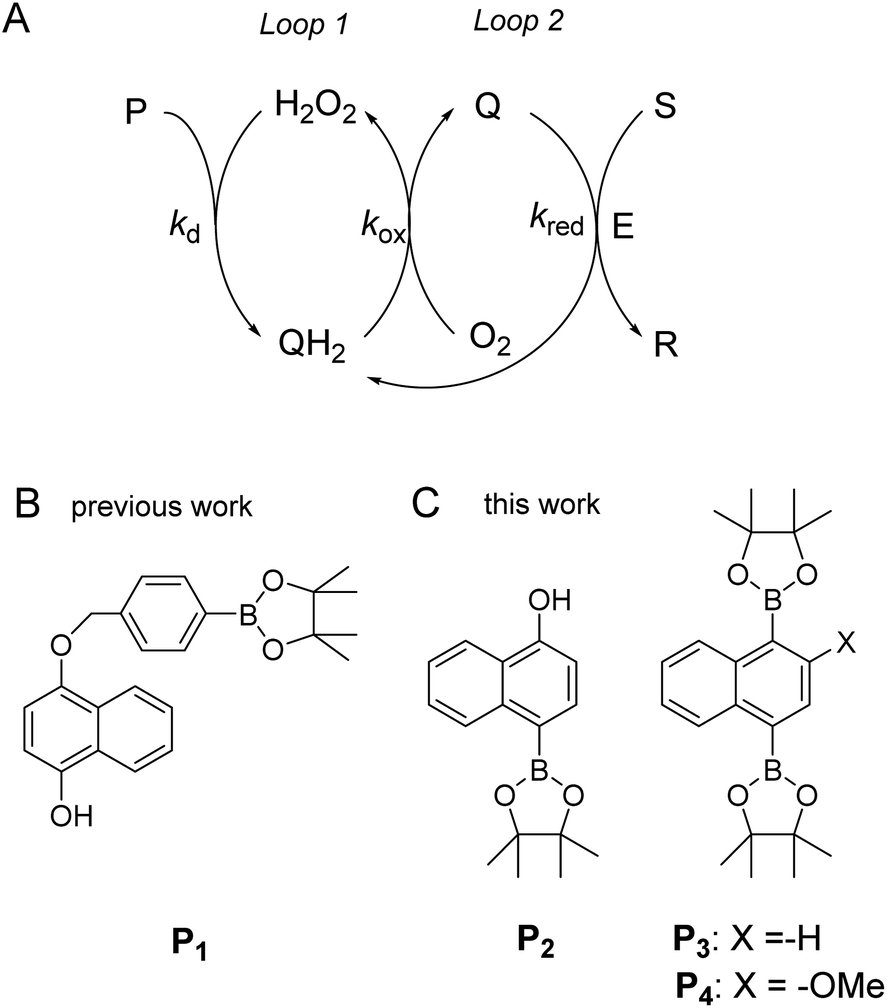
Exponential amplification by redox cross-catalysis and unmasking of doubly protected molecular probes - Chemical Science (RSC Publishing) DOI:10.1039/D1SC06086D

Non-innocent electrophiles go beyond Research Communities by Springer Nature

Base-Catalyzed Aryl-B(OH)2 Protodeboronation Revisited: From Concerted Proton Transfer to Liberation of a Transient Aryl Anion

Potassium Trimethylsilanolate Enables Rapid, Homogeneous Suzuki-Miyaura Cross-Coupling of Boronic Esters. - Abstract - Europe PMC

Base-Catalyzed Aryl-B(OH)2 Protodeboronation Revisited: From Concerted Proton Transfer to Liberation of a Transient Aryl Anion

PDF) Mechanism of Cu-Catalyzed Aryl Boronic Acid Halodeboronation Using Electrophilic Halogen: Development of a Base-Catalyzed Iododeboronation for Radiolabeling Applications

Base-Catalyzed Aryl-B(OH)2 Protodeboronation Revisited: From Concerted Proton Transfer to Liberation of a Transient Aryl Anion

Table 1 from Base-Catalyzed Aryl-B(OH)2 Protodeboronation Revisited: From Concerted Proton Transfer to Liberation of a Transient Aryl Anion.

Development and Molecular Understanding of a Pd‐Catalyzed Cyanation of Aryl Boronic Acids Enabled by High‐Throughput Experimentation and Data Analysis - De Jesus Silva - 2021 - Helvetica Chimica Acta - Wiley Online Library

Base-Catalyzed Aryl-B(OH)2 Protodeboronation Revisited: From Concerted Proton Transfer to Liberation of a Transient Aryl Anion











:max_bytes(150000):strip_icc()/scallops-grill-2000-40fcc03b0ead417aae52d482611b4552.jpg)



